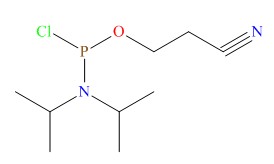
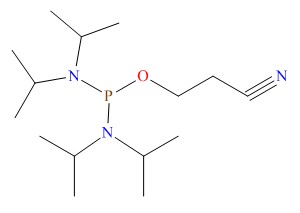
General Administration of medical devices issued a priority examination and approval procedures
Home >> Information >> Detailed content
Product list
Recommended products
news
Contact us
Q Q:27938229
Mobile phone:15981919713
Telephone:0371-56768238
Fax:0371-66658238
Email:chemprize@gmail.com
Mobile phone:15981919713
Telephone:0371-56768238
Fax:0371-66658238
Email:chemprize@gmail.com
General Administration of medical devices issued a priority examination and approval procedures
Time:2016-01-26 15:06:36 Click: author:chemprize
In order to further implement the State Council on the reform of drug and medical device review and approval system, security needs medical clinic, the State Food and drug administration through in-depth research and solicit opinions, recently released a "medical device" prior approval procedures, implemented since January 1, 2017.